
Areas of expertise
Medicinal chemistry , Pharmacological innovation
- Professor
- Scientific co-head of the Nuclear Magnetic Resonance Spectroscopy Laboratory
Armand-Frappier Santé Biotechnologie Research Centre
531 des Prairies Blvd.
Laval, Quebec H7V 1B7
CANADA
Research in my laboratory is centered around discovering drugs, practical tools and interesting chemical properties. Our goal is to explore the initial and essential steps of drug discovery – finding the starting chemical matter or seeds for designing future drugs that target diseases (Fig. 1). We are developing new breakthrough technologies that involve fragment-based lead discovery (FBLD) for finding these drug seeds. FBLD involves the screening of small libraries of low molecular-weight compounds called ”fragments” to search for new binders/inhibitors that target pathogens or specific proteins or RNA. These inhibitors can then be employed as scaffolds from which appendages are systematically added to improve potency and to “build a drug”.
However, serious issues remain that limit the practical application of FBLD. My laboratory aims to overcome these limitations and thus expediate the discovery of quality starting chemical-matter for any drug discovery program. This is happening by exploiting the team’s “decades of experience in pharma” and by evaluating and re-engineering each step along the FBLD pathway. We are implementing a synergistic complement of biology, biophysics and chemistry to overcome historic barriers. For example, hardware and software automation is being introduced at keys steps, and new medicinal chemistry and affinity NMR Kd cycles are being implemented to facilitate the transformation of binders to drug leads.
Developing and implementing these strategies requires a network of people, expertise and technologies. This is being achieved via the establishment of the new Drug Discovery Network (DDN). Teams of researchers from the University Quebec INRS-IAF, Harvard Medical School, Broad Institute, NMX and Key Organics are developing new, well-curated fragment libraries of fragments ready for high-throughput screening purposes. New NMR-based target-screening strategies have been introduced and successfully applied with many biotechnology companies located in Boston, Montreal and San Francisco (Fig. 2) to target cancer, cardiovascular diseases and infections by pathogens. The complexities of analyzing massive datasets are being simplified by the design of in-house software (Fig. 2). Interestingly, we also launched phenotype fragment screens (Fig. 3) and discovered that this strategy surprisingly provides selective inhibitors against pathogens such as Leishmania, Neisseria meningitidis, and those causing malaria and tuberculosis. These techniques are now being used in laboratories worldwide via the DDN and the International Pasteur Institute Network.
[caption id="attachment_24951" align="alignnone" width="710"] Fig. 1. Overview of the project for re-engineered the processes for fragment-based lead-discovery. A multidisciplinary team is currently improving FBLD strategies.[/caption]
[caption id="attachment_24953" align="alignnone" width="558"]
Fig. 1. Overview of the project for re-engineered the processes for fragment-based lead-discovery. A multidisciplinary team is currently improving FBLD strategies.[/caption]
[caption id="attachment_24953" align="alignnone" width="558"] Fig. 2. Fragment hits identified by consensus DLBS/STD/T2-CPMG NMR screening. New software developed in-house. An example of an X-ray structure solved for a fragment-protein complex. Multiple screens are currently underway to target diseases such as cancer. Click on the image to enlarge it.[/caption]
[caption id="attachment_24957" align="alignnone" width="535"]
Fig. 2. Fragment hits identified by consensus DLBS/STD/T2-CPMG NMR screening. New software developed in-house. An example of an X-ray structure solved for a fragment-protein complex. Multiple screens are currently underway to target diseases such as cancer. Click on the image to enlarge it.[/caption]
[caption id="attachment_24957" align="alignnone" width="535"] Fig. 3. Targeting Leishmania parasite by phenotype fragment screening. Cell-culture active fragment MAGA 005 was discovered. Click on the image to enlarge.[/caption]
Fig. 3. Targeting Leishmania parasite by phenotype fragment screening. Cell-culture active fragment MAGA 005 was discovered. Click on the image to enlarge.[/caption]
 Fig. 1. Overview of the project for re-engineered the processes for fragment-based lead-discovery. A multidisciplinary team is currently improving FBLD strategies.[/caption]
[caption id="attachment_24953" align="alignnone" width="558"]
Fig. 1. Overview of the project for re-engineered the processes for fragment-based lead-discovery. A multidisciplinary team is currently improving FBLD strategies.[/caption]
[caption id="attachment_24953" align="alignnone" width="558"] Fig. 2. Fragment hits identified by consensus DLBS/STD/T2-CPMG NMR screening. New software developed in-house. An example of an X-ray structure solved for a fragment-protein complex. Multiple screens are currently underway to target diseases such as cancer. Click on the image to enlarge it.[/caption]
[caption id="attachment_24957" align="alignnone" width="535"]
Fig. 2. Fragment hits identified by consensus DLBS/STD/T2-CPMG NMR screening. New software developed in-house. An example of an X-ray structure solved for a fragment-protein complex. Multiple screens are currently underway to target diseases such as cancer. Click on the image to enlarge it.[/caption]
[caption id="attachment_24957" align="alignnone" width="535"]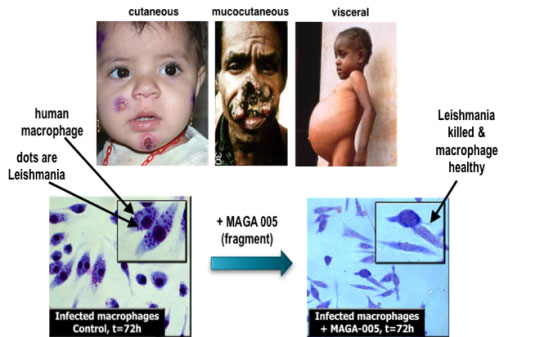 Fig. 3. Targeting Leishmania parasite by phenotype fragment screening. Cell-culture active fragment MAGA 005 was discovered. Click on the image to enlarge.[/caption]
Fig. 3. Targeting Leishmania parasite by phenotype fragment screening. Cell-culture active fragment MAGA 005 was discovered. Click on the image to enlarge.[/caption]
